Evaluation of the effect of ultrasonic waves on nitrate removal from aqueous solutions using Zinc- and Iron- coated activated carbon
-
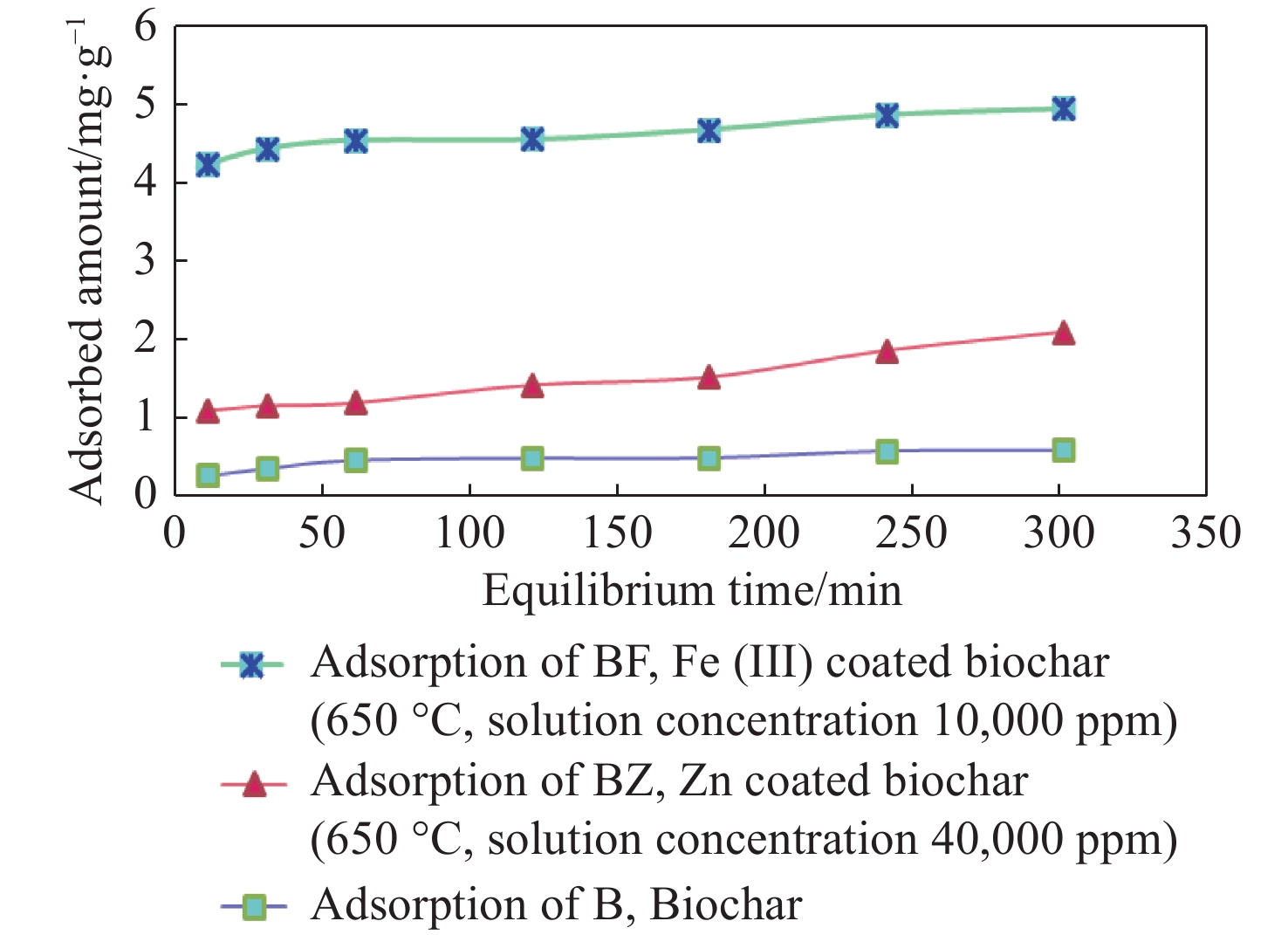
Abstract: Nitrate contamination in water represents a significant threat to both public health and the environment. While ultrasonic technology has emerged as an eco-friendly approach with potential for enhancing nitrate removal, its full capacity remains underexplored. This study aims to assess the effectiveness of ultrasound in improving nitrate removal from aqueous solutions using biochar derived from rice straw, modified with Fe3+ and Zn2+ as cationic bridges. Conducted at the water quality laboratory of Sari Agricultural Sciences and Natural Resources University, the experiments revealed that iron-coated biochar treatments (BF and BFU) exhibited outstanding performance in nitrate removal. Ultrasound application significantly enhanced nitrate removal efficiency, with the combination of ultrasonic waves and iron-coated biochar (BFU) achieving a maximum adsorption capacity (qm) of 3.664 mg/g, which surpassed non-sonicated treatments (BF: 3.345 mg/g) and reduced equilibrium time by 92% (from 60 min to 5 min). Furthermore, ultrasonic treatment improved the performance of Zn2+-coated biochar (BZU), boosting removal rates by more than 25% through cavitation-induced particle fragmentation and enhanced mass transfer. Mechanistic analysis indicated that ultrasound facilitates the homogenization of the adsorption surface, favoring Langmuir-type monolayer adsorption (R2 > 0.95), while the cationic bridges (Fe3+/Zn2+) strengthened electrostatic interactions with nitrate ions. Under optimized conditions, the combination of ultrasound and cation-modified biochar achieved over 90% nitrate removal, presenting a promising, energy-efficient, and sustainable solution for water treatment. These findings demonstrate the potential of ultrasonic-assisted, cation-modified biochar as a highly effective strategy for mitigating nitrate contamination in water systems.
-
Key words:
- Biochar /
- Iron coating /
- Langmuir model /
- Water quality /
- Zinc coating
BFU: Biochar coated with Fe3+ combined with ultrasonic treatment
U: Ultrasonic without adsorbent
BZU: Biochar coated with Zn2+ combined with ultrasonic treatment
BZ: Biochar coated with Zn2+
B: Treatments using biochar adsorbent
BF: Biochar coated with Fe3+
注释:1) List of nonstandard abbreviations: -
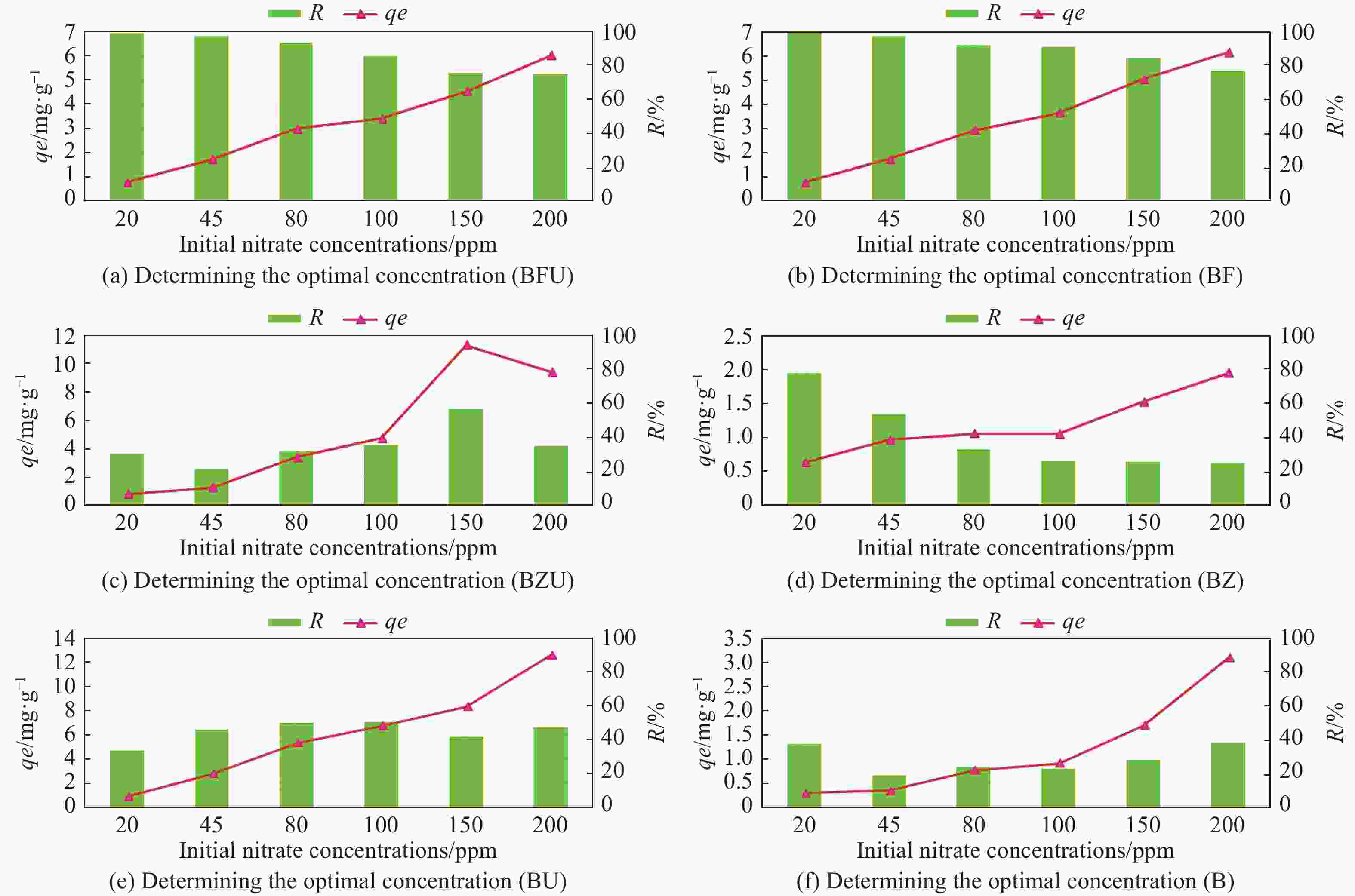
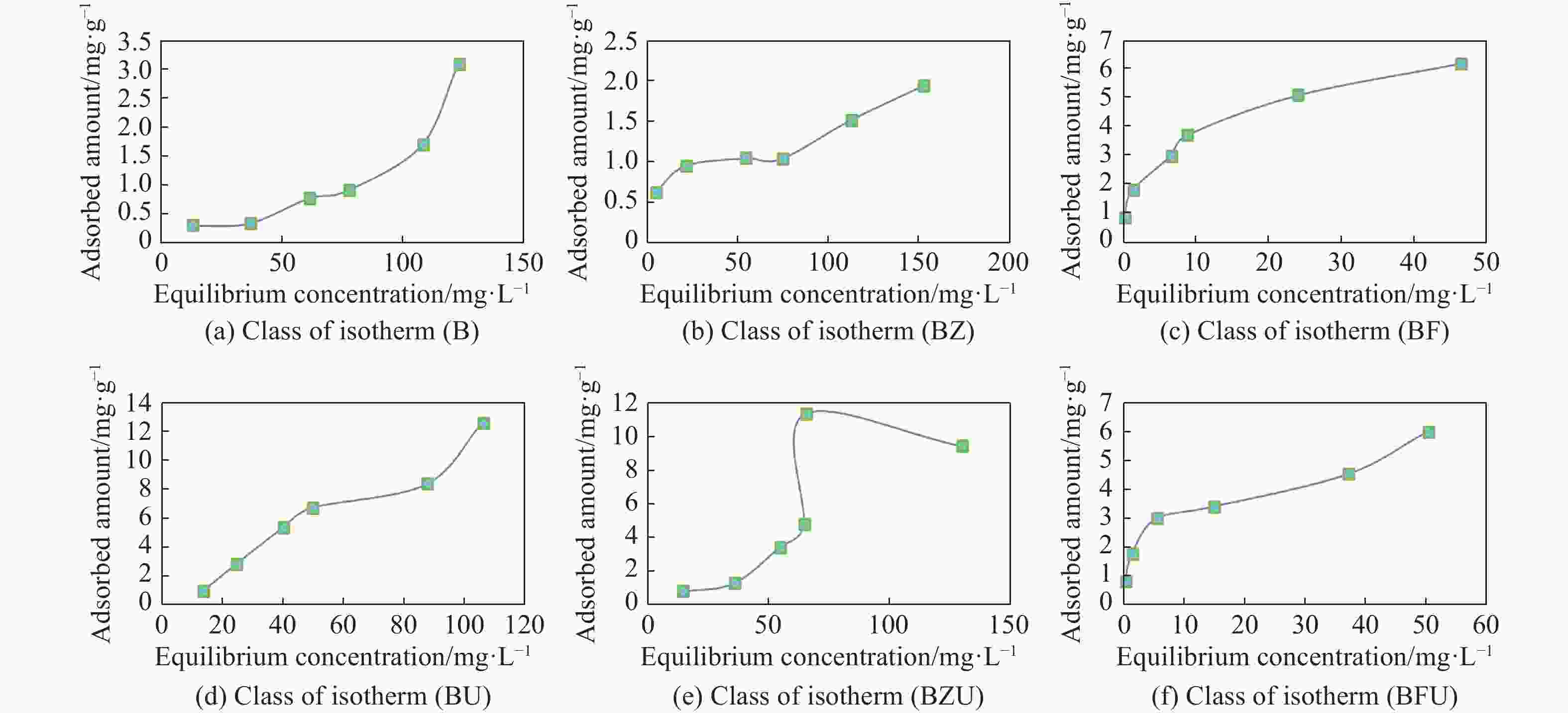
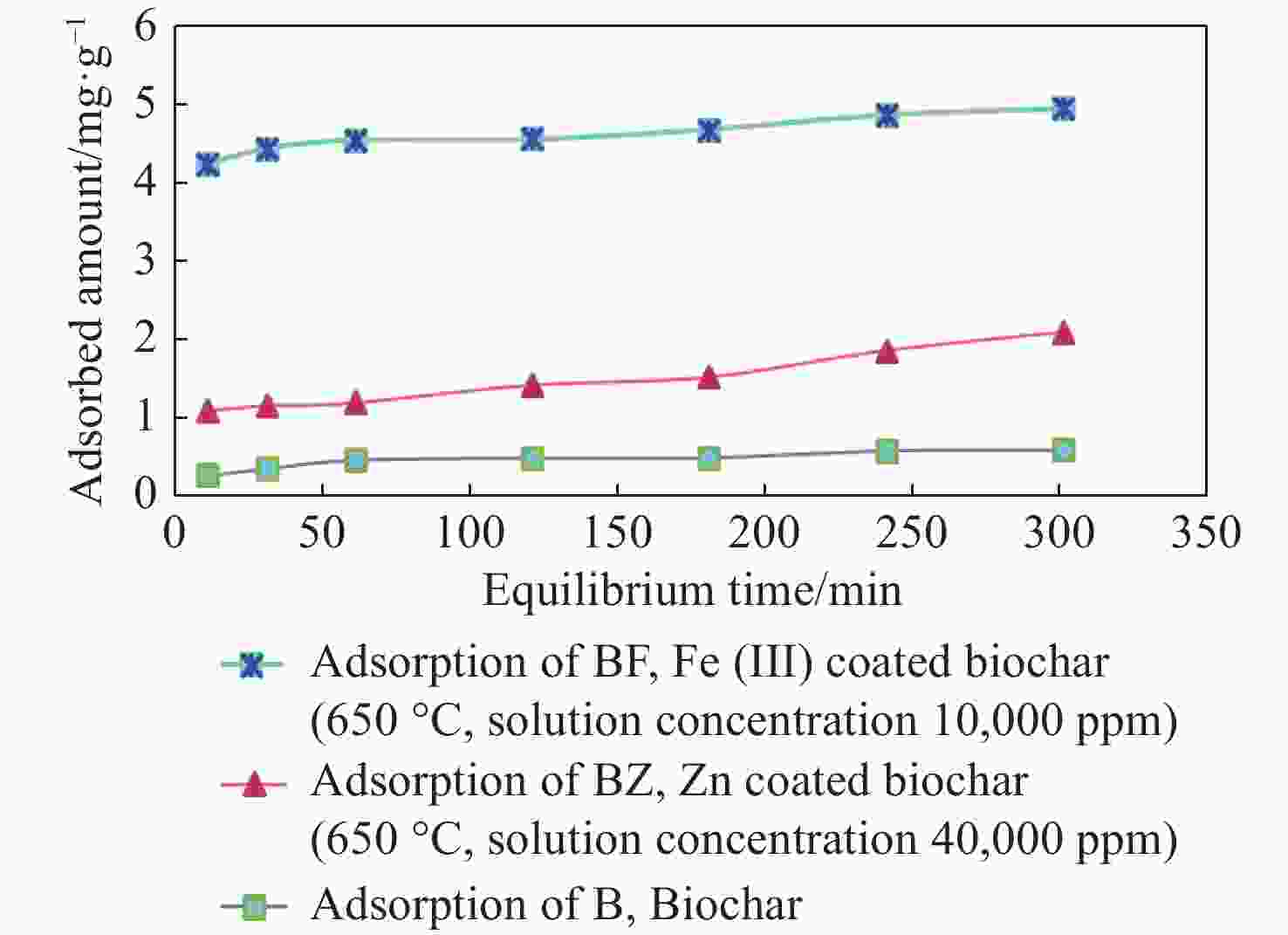
Table 1. Nitrate adsorption values in 6 different concentrations by experimental treatment
Initial
concentration
(mg/L)BF BFU BZ BZU B BU Ce** (mg/L) qe*
(mg/g)Ce (mg/L) qe (mg/g) Ce (mg/L) qe (mg/g) Ce (mg/L) qe (mg/g) Ce (mg/L) qe (mg/g) Ce (mg/L) qe (mg/g) 20 0.03 0.79 0.19 0.79 4.48 0.62 13.81 0.82 12.48 0.30 13.24 0.90 45 1.16 1.75 1.36 1.74 21.14 0.95 35.28 1.29 36.43 0.34 24.28 2.76 80 6.40 2.94 5.27 2.98 53.87 1.04 54.21 3.43 60.74 0.77 39.98 5.33 100 8.57 3.65 14.73 3.41 74.00 1.03 64.18 4.77 77.20 0.91 49.67 6.71 150 23.78 5.04 36.81 4.52 112.06 1.51 64.71 11.37 107.75 1.68 87.49 8.33 200 46.14 6.15 49.96 6.00 151.51 1.93 129.02 9.46 122.79 3.08 105.88 12.54 Notes: qe* is the amount of nitrate adsorbed per unit weight of biochar
Ce** is the equilibrium concentration of nitrate in solutionTable 2. The values obtained for Langmuir and Freundlich isotherm parameters and the correlation coefficient of nitrate adsorbed in different treatments
Isotherms parameters Treatment Langmuir Freundlich qm b R2 kf nf R2 BF 3.345 8.491 0.894 1909.414 3.641 0.985 BFU 3.664 1.405 0.95 1484.22 2.942 0.981 BZ 1.341 0.184 0.806 386.63 3.562 0.856 BZU - - 0.886 26.24 0.795 0.799 B 1.632 0.016 0.713 18.488 1.047 0.806 BU - - 0.946 56.48 0.852 0.944 Table 3. Percentage of nitrate removal in different treatments and experiments
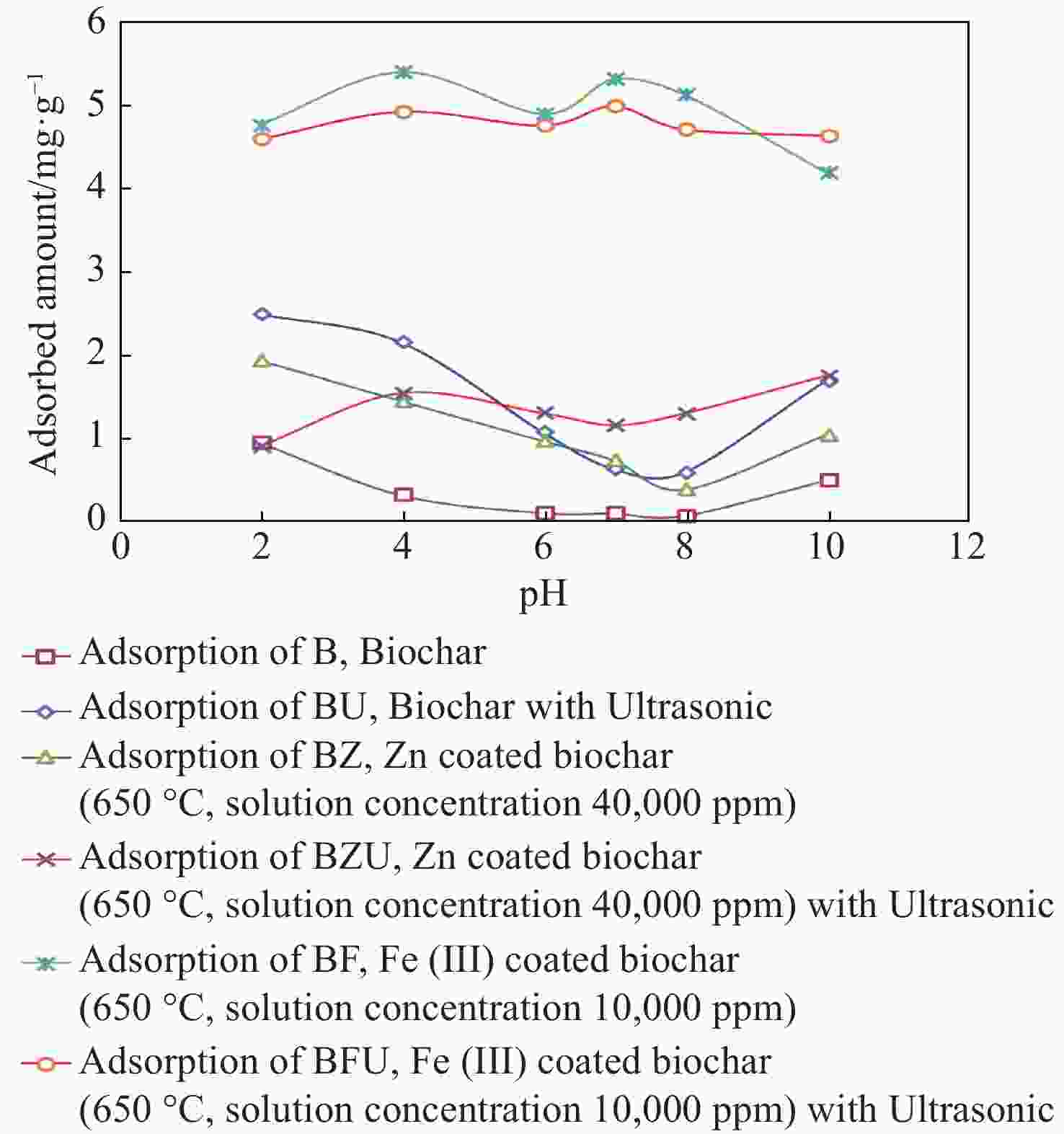
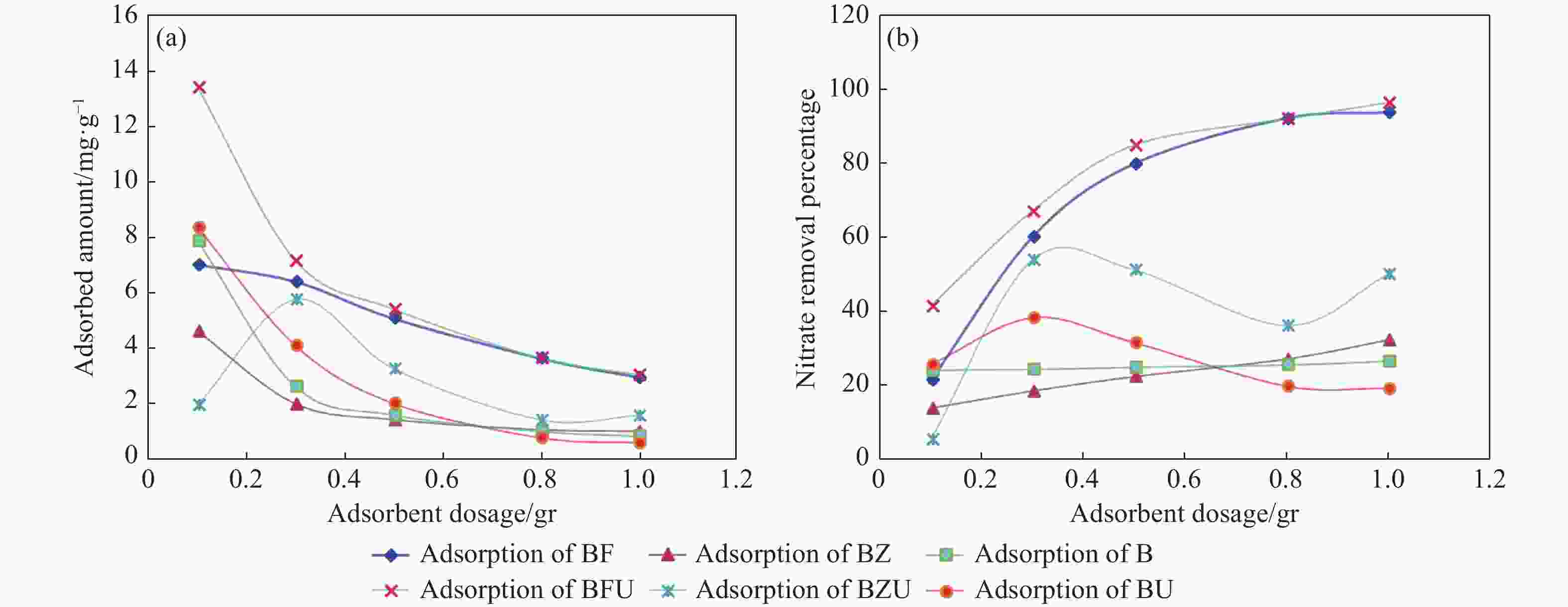
Nitrate removal percentage pH BF BFU BZ BZU B BU 2 74.39 71.70 30.19 13.99 14.75 38.91 4 84.37 76.94 22.55 24.06 4.82 33.58 6 76.49 74.30 15.03 20.29 1.50 16.67 7 82.97 77.96 11.46 18.01 1.40 9.79 8 80.11 73.59 5.91 20.23 0.96 9.15 10 65.49 72.42 16.18 27.34 7.61 26.43 Adsorbent Dosage (gr) 0.1 22.11 42.12 14.58 6.239 24.79 26.31 0.3 60.57 67.65 19.20 54.46 24.96 38.87 0.5 80.19 85.26 23.04 51.77 25.53 32.19 0.8 92.42 92.34 27.77 36.76 26.16 20.48 1 94.02 96.72 32.90 50.59 27.18 19.91 Initial nitrate concentrations 20 99.81 99.04 77.55 30.90 37.58 33.76 45 97.40 96.95 53.01 21.59 19.02 46.03 80 91.99 93.41 32.66 32.23 24.06 50.01 100 91.42 85.26 25.99 35.81 22.79 50.32 150 84.14 75.45 25.29 56.85 28.16 41.67 200 76.92 75.01 24.24 35.48 38.60 47.05 -
Alashti MR, Khoshravesh M, Sadegh-zadeh F, et al. 2024. The effect of ultrasonic waves on nitrate removal in aqueous solution. Iranian Journal of Irrigation and Drainage, 2(18): 319−328. Amininejad M, Boroomand-Nasab S, Moazed H, et al. 2019. Evaluation of nitrate removal from aqueous solution by nanostructure of Conocarpus. Scientific Research Journal of Irrigation and Water Engineering of Iran, 37: 167−180. DOI: 10.22125/iwe.2019.95882. APHA. 2012. Standard Methods for the Examination of Water and Wastewater. 22nd Edition, American Public Health Association, Washington DC. Cataldo DA, Maroon M, Schrader LE, et al. 1975. Rapid colorimetric determination of nitrate in plant tissues by nitration of salicylic acid. Communication of Soil Science and Plant Analysis, 6(1): 71−80. DOI: 10.1080/00103627509366547. Dehghani MH, Karri RR, Koduru JR, et al. 2023. Ultrasonic cavitation: Tackling organic pollutants in wastewater. Ultrasonics Sonochemistry, 94: 106302. Eissa R, Jeyakumar L, McKenzie DB, et al. 2024. Influence of biochar feedstocks on nitrate adsorption capacity. Earth, 5(4): 1080−1096. DOI: 10.3390/earth5040055. González-López J, Hu X, Rajahmundry R, et al. 2021. Critical evaluation of isotherm modeling in nitrate adsorption studies. Water, Air, and Soil Pollution, 232(7): 1−15. DOI: 10.1007/s11270-021-05345-6. Guo F, Zhang Y, Li H. 2023. The removal of ammonia-nitrogen from aquaculture water based on cavitation effect of ultrasonic vibration. Journal of Vibroengineering, 25(5): 1012−1020. DOI: 10.21595/vp.2023.23575extrica.com. Homagai PL, Poudel R, Paudyal H, et al. 2023. Adsorption of nitrate and nitrite anion by modified maize stalks from aqueous solutions. Environmental Science and Pollution Research, 30: 54682−54693. DOI: 10.1007/s11356-023-26179-y. Hosseingholilu B, Banakar A, Mostafaei M. 2019. Design and evaluation of a novel ultrasonic desalination system by response surface methodology. Desalination and Water Treatment, 164: 263−275. DOI: 10.5004/dwt.2019.24458. Hu Q, Chen N, Feng C, et al. 2015. Nitrate adsorption from aqueous solution using granular chitosan-Fe3+ complex. Journal of Applied Surface Science, 374: 1−9. DOI: 10.1016/j.apsusc.2015.04.049. Janajreh I, Ali U, Hawwa M. 2022. Sonicated direct contact membrane distillation: Influence of sonication parameters. Desalination, 533: 115779. DOI: 10.1016/j.desal.2022.115779. Kumar R, Singh R. 2024. Removal of nitrate ion from aqueous solution using palmyrah nut shell activated carbon: Factorial optimization and equilibrium studies. Discover Civil Engineering, 2(1): 54. DOI: 10.1007/s44290-024-00054-2. Lahjouj A, Hmaidi AE, Bouhafa K. 2020. Spatial and statistical assessment of nitrate contamination in groundwater: Case of Sais Basin, Morocco. Journal of Groundwater Science and Engineering, 8(2): 143−157. DOI: 10.19637/j.cnki.2305-7068.2020.02.006. Mohamad Aris KH, Ramli S, Othman Z, et al. 2018. Evaluation of ammonia-nitrogen removal by ultrasonic irradiation in synthetic solution using response surface methodology. Key Engineering Materials, 797: 108−117. DOI: 10.4028/www.scientific.net/KEM.797.108. Mood SH, Pelaez-Samaniego MR, Han Y, et al. 2024. Iron- and nitrogen-modified biochar for nitrate adsorption from aqueous solution. Sustainability, 16(13): 5733. DOI: 10.3390/su16135733. Nguyen Le KT, Maldonado JFG, Nguyen TL, et al. 2024. The short-term effect of nitrogen on freshwater cyanobacteria and cyanotoxins. Frontiers in Water, 6: 1432183. DOI: 10.3389/frwa.2024.1432183. Ogata F, Imai D, Kawasaki N. 2015. Adsorption of nitrate and nitrite ions onto carbonaceous material produced from soybean in a binary solution system. Journal of Environmental Chemical Engineering, 3: 155−161. DOI: 10.1016/j.jece.2014.11.025. Oprescu EE, Enascuta EC, Vasilievici G, et al. 2022. Preparation of magnetic biochar for nitrate removal from aqueous solutions. Reaction Kinetics, Mechanisms and Catalysis, 135(3): 1647−1665. DOI: 10.1007/s11144-022-02263-1. Rashwan SS, Dincer I, Mohany A. 2020. An investigation of ultrasonic based hydrogen production. International Journal of Hydrogen Energy, 205: 118006. DOI: 10.1016/j.energy.2020.118006. Reusch TBH, Dierking J, Andersson HC, et al. 2018. The Baltic Sea as a time machine for the future coastal ocean. Science Advances, 4: 8195. DOI: 10.1126/sciadv.aar8195. Serna-Galvis EA, Porras J, Torres-Palma RA. 2022. A critical review on the sonochemical degradation of organic pollutants in urine, seawater, and mineral water. Ultrasonics Sonochemistry, 82: 105861. DOI: 10.1016/j.ultsonch.2021.105861. Sharma A, Gupta S. 2024. Parthenium hysterophorus-derived iron-coated biochar: A sustainable solution for nitrate and phosphate removal from water. Biomass Conversion and Biorefinery, 14(3): 821. DOI: 10.1007/s13399-024-05821-w. Ward MH, Jones RR, Brender JD, et al. 2024. Drinking-water nitrate and human health: An updated review. Environmental Science & Technology, 58(4): 2345−2357. Zameni L, Sadegh-zadeh F, Seh-Bardan BJ. 2016. Nitrate leaching in soil modified by biochar and iron coated biochar. M.S. thesis, Sari Agricultural Sciences and Natural Resources University. Zhang M, Song G, Gelardi DL, et al. 2020. Evaluating biochar and its modifications for the removal of ammonium, nitrate, and phosphate in water. Water Research, 186: 116303. DOI: 10.1016/j.watres.2020.116303. -
 E-mail alert
E-mail alert Rss
Rss


 下载:
下载: